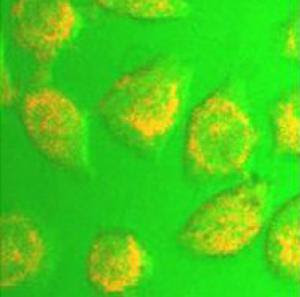
 Quantum dots apart from being neat things in and of themselves-could be put to some very interesting uses. The first application of quantum dots will be for biological labels used in medical imaging. Researchers tag proteins and nucleic acids with quantum dots. When they shine ultraviolet ray on the sample, the quantum dots glow at a specific wavelength and indicate the locations of attached proteins. Quantum dots have advantages over materials currently used for this application. For example they glow longer.
Quantum dots apart from being neat things in and of themselves-could be put to some very interesting uses. The first application of quantum dots will be for biological labels used in medical imaging. Researchers tag proteins and nucleic acids with quantum dots. When they shine ultraviolet ray on the sample, the quantum dots glow at a specific wavelength and indicate the locations of attached proteins. Quantum dots have advantages over materials currently used for this application. For example they glow longer.Researchers are also hoping that quantum dots could eventually provide energy efficient lighting for general use, in your house, office or neighborhood street lamps. In these applications a light emitting diode (LED) or other source of UV light would shine on quantum dots, which would then light up. By mixing different sizes (and the associated colors) of quantum dots together, you could generate white light. Generating light from quantum dots would work like generating fluorescent light but without the bulky fluorescent tube. This method would also avoid the wasted heat that you get with your typical incandescent light bulb.
Passing an electrical current through an LED also generates light. A company called Q vision is attempting to use techniques developed MIT to design a quantum dot LED: A layer of quantum dots sandwiched between conductive organic layers. Passing a c
Flat panel TV displays using quantum dots LEDs may provide more vibrant colors than current flat panel displays based on liquid crystals Display (LCD) Technology.
urrent through the dots generates light